How Do You Know Which Way an Electron or Atom Will Point to Electric Fields
Electric Charge in the Atom
Atoms contain negatively charged electrons and positively charged protons; the number of each determines the cantlet's cyberspace charge.
Learning Objectives
Identify factors that determine the atom's net charge
Key Takeaways
Key Points
- A proton is a positively charged particle located in the nucleus of an atom. An electron has [latex]\frac{ane}{1836}[/latex] times the mass of a proton, but an equal and opposite negative accuse.
- An elementary charge — that of a proton or electron — is approximately equal to 1.6×10-19Coulombs.
- Different protons, electrons can move from cantlet to cantlet. If an atom has an equal number of protons and electrons, its net charge is 0. If it gains an extra electron, it becomes negatively charged and is known equally an anion. If it loses an electron, it becomes positively charged and is known every bit a cation.
Key Terms
- nucleus: the massive, positively charged primal part of an atom, made upward of protons and neutrons
Overview of Atomic Electrical Charges
Atoms, the primal building blocks of all molecules, consist of three types of particles: protons, neutrons, and electrons. Of these 3 subatomic particle types, two (protons and electrons) bear a net electric accuse, while neutrons are neutral and take no net charge.
Both protons and electrons have charge that is quantized. That is, the magnitude of their corresponding charges, which are equal each other, is 1. This standard value is equal to approximately 1.six×10-xix Coulombs.
Protons
Protons are plant in the center of the atom; they, with neutrons, brand up the nucleus. Protons accept a accuse of +one and a mass of 1 atomic mass unit, which is approximately equal to 1.66×10-24 grams. The number of protons in an atom defines the identity of the element (an atom with ane proton is hydrogen, for example, and an cantlet with two protons is helium). Every bit such, protons are relatively stable; their number rarely changes, only in the case of radioactive disuse.
Electrons
Electrons are found in the periphery of the atom and have a charge of -1. They are much smaller than protons; their mass is [latex]\frac{one}{1836}[/latex] amu. Typically in modeling atoms, protons and neutrons are regarded as stationary, while electrons move virtually in the space exterior the nucleus like a deject. The negatively charged electronic cloud indicates the regions of the infinite where electrons are likely to be institute. The electrons cloud patterns are extremely circuitous and is of no importance to the discussion of electric charge in the atom. More important is the fact that electrons are labile; that is, they tin exist transferred from one cantlet to the adjacent. It is through electronic transfer that atoms become charged.
Ions
In the ground state, an atom will have an equal number of protons and electrons, and thus volition have a net charge of 0. However, because electrons tin exist transferred from one atom to another, it is possible for atoms to become charged. Atoms in such a state are known as ions.
If a neutral atom gains an electron, it becomes negative. This kind of ion is called an anion.
If a neutral atom loses an electron, it becomes positive. This kind of ion is called a cation.
The steady flow of electrons is called current. Current is what flows through electrical wires and powers electronics items, from light bulbs to televisions.
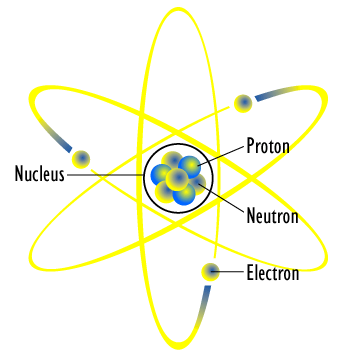
Planetary Model of an Atom: Small electrons orbit the large and relatively fixed nucleus of protons and neutrons.
Properties of Electric Charges
Electrical charge is a fundamental physical property of matter that has many parallels to mass.
Learning Objectives
Draw properties of electric charge, such as its relativistic invariance and its conservation in closed systems
Primal Takeaways
Key Points
- Charge is measured in Coulombs (C), which represent 6.242×tenxviii e, where east is the charge of a proton. Charges tin can exist positive or negative, and every bit such a singular proton has a charge of i.602×10−xix C, while an electron has a charge of -one.602×10−19 C.
- Electric charge, like mass, is conserved. The strength generated past ii charges is of the same form as that generated past two masses and, like gravity, forcefulness from an electrical field is both conservative and primal.
- Electric charge is a relativistic invariant. That is, accuse (unlike mass) is independent of speed. Whereas the mass of a particle will exponentially rise as its speed approaches that of low-cal, charge will remain constant.
Key Terms
- coulomb: In the International System of Units, the derived unit of measurement of electric charge; the amount of electric charge carried by a electric current of 1 ampere flowing for 1 second. Symbol: C
- gravity: Resultant force on World's surface, of the allure by the Earth's masses, and the centrifugal pseudo-forcefulness caused by the Globe's rotation.
- electric field: A region of space effectually a charged particle, or between two voltages; it exerts a force on charged objects in its vicinity.
Properties of Electrical Charge
Electric accuse, like mass and volume, is a concrete property of matter. Its SI unit is known as the Coulomb (C), which represents 6.242×1018 e, where east is the accuse of a proton. Charges can exist positive or negative; a singular proton has a charge of 1.602×10−nineteen C, while an electron has a accuse of -1.602×10−19 C.
Invariance
Like mass, electric charge in a airtight arrangement is conserved. As long as a system is impermeable, the amount of charge within it will neither increase nor decrease; it tin only be transferred. All the same, electric accuse differs from other properties—like mass—in that it is a relativistic invariant. That is, accuse is contained of speed. The mass of a particle will rise exponentially as its speed approaches that of light, its charge, however, will remain constant.
The independence of electrical charge from speed was proven through an experiment in which ane fast-moving helium nucleus (two protons and two neutrons bound together) was proven to have the same charge as two separate, tiresome-moving deuterium nuclei (i proton and one neutron leap together in each nucleus).
Attraction and Repulsion
Electric accuse is a property that produces forces that tin can attract or repel matter. Mass is similar, although it can just concenter thing, not repel it. Still, the formula describing the interactions between charges is remarkably similar to that which characterizes the interactions betwixt masses. For electric fields, the strength (F) is related to the charges (q1, q2) and the distance (r) between them as:
[latex]\text{F}=\frac{i}{four\pi \epsilon_0}\frac {\text{q}_1\text{q}_2}{\text{r}^two}[/latex]
where π and [latex]\epsilon_0[/latex] are constants. This is known as Coulomb'south Law.

Coulomb'due south Law: The forces (Fi and Fii) sum to produce the total strength, which is calculated by Coulomb's Law and is proportional to the product of the charges qi and q2, and inversely proportional to the square of the distance (r21) between them.
The formula for gravitational strength has exactly the same form equally Coulomb's Law, simply relates the product of two masses (rather than the charges) and uses a unlike constant. Both act in a vacuum and are fundamental (depend simply on distance between the forces) and conservative (independent of path taken). However, it should be noted that when comparison like terms, accuse-based interaction is substantially greater than that based on mass. For example, the electrical repulsion between two electrons is about 1042 times stronger than their gravitational attraction.
Accuse Separation
Charge separation, oftentimes referred to as static electricity, is the building of space betwixt particles of reverse charges.
Learning Objectives
Identify factors that can create charge separation
Primal Takeaways
Key Points
- Considering electrons are labile (i.eastward., they can be transferred from atom to atom), it is possible for "charge separation" to occur. This phenomenon is oft commonly referred to as static electricity.
- Charge separation can be created by friction, pressure, heat, and other charges.
- Charge separation tin can accomplish a disquisitional level, whereat it is discharged. Lightning is a common example.
Central Terms
- discharge: the act of releasing an accumulated charge
- static electricity: an electric accuse that has built upward on an insulated body, often due to friction
- nucleus: the massive, positively charged key part of an atom, fabricated upward of protons and neutrons
All thing is equanimous of atoms made upwards of negatively-charged electrons and positively-charged protons. In the ground state, each cantlet is of neutral accuse—its protons and electrons are equal in number, and it exists with no permanent dipole. Because electrons are labile (i.e., they tin be transferred from atom to atom) it is possible for the phenomenon of "charge separation" (often referred to as static electricity) to occur.

Static Electricity: Due to friction between her pilus and the plastic slide, the daughter on the left has created accuse separation, resulting in her pilus being attracted to the slide.
In chemistry, this accuse separation is illustrated simply past the transfer of an electron from i atom to another equally an ionic bail is formed. In physics, at that place are many other instances of charge separation that cannot exist written as formal chemical reactions. Consider, for instance, rubbing a airship on your pilus. In one case yous pull the balloon away, your hair volition stand on stop and "reach" towards the balloon. This is considering electrons from ane have transferred to the other, causing 1 to exist positive and the other to be negative. Thus, the contrary charges attract. A similar example tin be seen in playground slides (as shown in ).
Accuse separation can be created non only past friction, but by pressure, heat, and other charges. Both pressure and rut increase the energy of a cloth and can crusade electrons to break free and separate from their nuclei. Accuse, meanwhile, can attract electrons to or repel them from a nucleus. For example, a nearby negative accuse can "push button" electrons away from the nucleus around which they typically orbit. Accuse separation occurs often in the natural world. It can have an extreme effect if information technology reaches a critical level, whereat it becomes discharged. Lightning is a common example.
Polarization
Dielectric polarization is the miracle that arises when positive and negative charges in a fabric are separated.
Learning Objectives
Identify two ways polarization can occur on the molecular level
Key Takeaways
Fundamental Points
- Dielectrics are insulators that are capable of beingness polarized by an electrical field. That is, their charges cannot flow freely, only can yet be induced to redistribute unevenly.
- Electric fields applied to atoms will push electrons away from the field. In the case of polar molecules, the negative ends thereof will marshal themselves away from the field while the positive ends will be towards the field.
- An instantaneous polarization occurs when ions, through natural, random vibrations, become distributed asymmetrically such that one area is more dense with i type of ion than another.
Key Terms
- dipole moment: The vector product of the accuse on either pole of a dipole and the distance separating them.
- dielectric: An electrically insulating or nonconducting material considered for its electric susceptibility (i.e., its property of polarization when exposed to an external electrical field).
- insulator: A substance that does not transmit rut (thermal insulator), audio (audio-visual insulator) or electricity (electrical insulator).
The concept of polarity is very broad and can exist applied to molecules, calorie-free, and electrical fields. For the purposes of this cantlet, we focus on its meaning in the context of what is known as dielectric polarization—the separation of charges in materials.
Dielectrics
A dielectric is an insulator that tin can be polarized by an electric field, meaning that it is a material in which charge does not menstruation freely, just in the presence of an electric field it can shift its charge distribution. Positive charge in a dielectric volition drift towards the practical field, while negative charges will shift away. This creates a weak local field within the textile that opposes the applied field.
Dissimilar materials volition react differently to an induced field, depending on their dielectric constant. This abiding is the degree of their polarizability (the extent to which they get polarized).
Diminutive Model
The most basic view of dielectrics involves because their charged components: protons and electrons. If an electrical field is applied to an cantlet, the electrons in the cantlet will migrate away from the practical field. The protons, however, remain relatively exposed to the field. This separation creates a dipole moment, as shown in.

Reaction of an Cantlet to an Applied Electric Field: When an electric field (E) is applied, electrons drift away from the field. Their average location is displaced from the average location of the protons (which hasn't moved) past a distance of d. The atom'due south dipole moment is represented by Chiliad.
Dipole Polarization
On the molecular level, polarization can occur with both dipoles and ions. In polar bonds, electrons are more than attracted to one nucleus than to the other. One example of a dipole molecule is water, (HtwoO), which has a bent shape (the H-O-H bending is 104.45°) and in which the oxygen pulls electron density abroad from the H atoms, leaving the H relatively positive and the O relatively negative, as shown in.

Water Molecule: Water is an example of a dipole molecule, which has a bent shape (the H-O-H bending is 104.45°) and in which the oxygen pulls electron density away from the H atoms, leaving the H relatively positive and the O relatively negative.
When a dipolar molecule is exposed to an electrical field, the molecule will align itself with the field, with the positive end towards the electric field and the negative terminate away from it.
Ionic Polarization
Ionic compounds are those that are formed from permanently charge-separated ions. For example, tabular array common salt (NaCl) is formed from Na+ and Cl– ions that are not formally bound to 1 another through a chemical bond, just collaborate very strongly due to their opposite charges.
Ions are nonetheless free from one another and will naturally move at random. If they happen to move in a mode that is asymmetrical, and results in a greater concentration of positive ions in one area and a greater concentration of negative ions in another, the sample of ionic compound volition exist polarized—a phenomenon is known as ionic polarization.
Static Electricity, Accuse, and the Conservation of Charge
Electric charge is a physical holding that is perpetually conserved in corporeality; it can build upwardly in matter, which creates static electricity.
Learning Objectives
Codify rules that apply to the creation and the destruction of electric accuse
Key Takeaways
Fundamental Points
- Electrical charge is a physical property of matter created by an imbalance in the number of protons and electrons in a substance.
- Accuse tin be created or destroyed. Yet, whatsoever creation or elimination of charge occurs at a ratio of 1:i between positive and negative charges.
- Static electricity is when an backlog of electric charge collects on an object's surface.
Key Terms
- electrical charge: A quantum number that determines the electromagnetic interactions of some subatomic particles; by convention, the electron has an electric charge of -1 and the proton +i, and quarks have fractional charge.
- discharge: the human activity of releasing an accumulated accuse
- static electricity: an electric charge that has built upwards on an insulated body, oft due to friction
Electric accuse is a physical belongings of matter. It is created by an imbalance in a substance's number of protons and electrons. The matter is positively charged if information technology contains more protons than electrons, and it is negatively charged if it contains more electrons than protons. In both instances, charged particles volition feel a force when in the presence of other charged thing.
Charges of like sign (positive and positive, or negative and negative) volition repel each other, whereas charges of contrary sign (positive and negative) will concenter each another, as shown in.

Accuse Repulsion and Allure: Charges of similar sign (positive and positive, or negative and negative) will repel each other, whereas charges of opposite sign (positive and negative) will attract each other.
The SI unit of measurement for charge is the Coulomb (C), which is approximately equal to [latex]6.24\times 10^{18}[/latex] unproblematic charges. (An elementary charge is the magnitude of accuse of a proton or electron. )
Conservation of Charge
Charge, like thing, is essentially abiding throughout the universe and over fourth dimension. In physics, charge conservation is the principle that electrical charge can neither be created nor destroyed. The net quantity of electric charge, the amount of positive charge minus the amount of negative charge in the universe, is always conserved.
For any finite volume, the police of conservation of accuse (Q) tin exist written every bit a continuity equation:
[latex]\text{Q}(\text{t}_2)=\text{Q}(\text{t}_1)+\text{Q}_{\text{in}}-\text{Q}_{\text{out}}[/latex]
where Q(t i) is the accuse in the organisation at a given time, Q(t 2) is the charge in the aforementioned system at a later fourth dimension, Qin is the charge that has entered the organization between the two times, and Qout is the corporeality of accuse that has left the system between the ii times.
This does not hateful that individual positive and negative charges cannot exist created or destroyed. Electric charge is carried past subatomic particles such every bit electrons and protons, which tin can be created and destroyed. For example, when particles are destroyed, equal numbers of positive and negative charges are destroyed, keeping the cyberspace amount of charge unchanged.
Static Electricity
Static electricity is when an excess of electric charge collects on an object's surface. Information technology tin exist created through contact between materials, a buildup of force per unit area or heat, or the presence of a charge. Static electricity can too exist created through friction between a airship (or another object) and human being hair (encounter ). It can be observed in storm clouds as a result of pressure buildup; lightning (encounter ) is the discharge that occurs after the charge exceeds a critical concentration.

Static Electricity: Due to friction between her pilus and the plastic slide, the girl on the left has created accuse separation, resulting in her pilus being attracted to the slide.

Lightning: Lightning is a dramatic natural case of static discharge.
Conductors and Insulators
Based on the power to conduct current, materials are divided into conductors and insulators.
Learning Objectives
Identify conductors and insulators among common materials
Key Takeaways
Key Points
- Resistivity, a physical property that measures the power of a material to carry electric current, is the main factor in determining whether a substance is a usher or an insulator.
- Conductors comprise electrical charges that, when exposed to a potential difference, move towards i pole or the other. This flow of charge is electric current.
- Insulators are materials in which the internal charge cannot menstruum freely, and thus cannot conduct electric current to an observable degree when exposed to an electric field.
Key Terms
- conductor: A material which contains movable electric charges.
- insulator: A substance that does non transmit heat (thermal insulator), sound (audio-visual insulator) or electricity (electrical insulator).
- resistivity: In general, the resistance to electrical current of a material; in particular, the degree to which a material resists the flow of electricity.
Overview
All materials tin can exist categorized as either insulators or conductors based on a physical property known as resistivity.
An insulator is a cloth in which, when exposed to an electrical field, the electric charges exercise not menstruation freely—it has a high resistivity. Conversely, a conductor is a material that permits the flow of electric charges in one or more directions—its resistivity is depression.
Conductors
All conductors incorporate electrical charges that, when exposed to a potential deviation, move towards 1 pole or the other. The positive charges in a conductor will migrate towards the negative stop of the potential departure; the negative charges in the material will motility towards the positive end of the potential difference. This flow of charge is electrical current.
Ionic substances and solutions can conduct electricity, simply the most common and effective conductors are metals. Copper is usually used in wires due to its high electrical conductivity and relatively inexpensive price. Nevertheless, gold-plated wires are sometimes used in instances in which especially high electrical conductivity is necessary.
Every usher has a limit to its ampacity, or amount of current it tin bear. This commonly is the current at which the heat released due to resistance melts the material.
Insulators
Insulators are materials in which the internal charge cannot flow freely, and thus cannot conduct electric current to an appreciable degree when exposed to an electric field.
While in that location is no perfect insulator with infinite resistivity, materials like glass, paper and Teflon have very high resistivity and can effectively serve as insulators in virtually instances.
But equally conductors are used to carry electrical current through wires, insulators are unremarkably used as blanket for the wires.
Insulators, like conductors, have their physical limits. When exposed to enough voltage, an insulator will experience what is known as electrical breakup, in which electric current suddenly spikes through the material every bit it becomes a conductor.

Conductor and Insulator in a Wire: This wire consists of a core of copper (a usher) and a coating of polyethylene (an insulator). The copper allows electric current to menstruation through the wire, while the polyethylene ensures that the current does not escape.
The Millikan Oil-Driblet Experiment
In 1911, using charged droplets of oil, Robert Millikan was able to determine the charge of an electron.
Learning Objectives
Explain the difference in value of a real electron's charge and the charge measured by Robert Millikan
Fundamental Takeaways
Key Points
- The Oil-Drop Experiment involved ionizing droplets of oil equally they savage through the air, and balancing the strength of gravity with the forcefulness of an electric field applied by electrodes above and below the droplet.
- Millikan could not direct count the number of electrons on each oil droplet, but found that the mutual denominator between all measured charges was equal to ane.5924(17)×ten−nineteen C, and thus concluded that this value was the charge of an electron.
- The measured value of an electron's charge, ane.5924(17)×ten−19 C, differs from the accepted value of one.602176487(40)×ten−19 C past less than ane percent.
Key Terms
- voltage: The amount of electrostatic potential between two points in space.
- terminal velocity: The speed at which an object in costless-fall and not in a vacuum ceases to accelerate downwards because the strength of gravity is equal and opposite to the drag force acting confronting it.
- electric field: A region of space around a charged particle, or between two voltages; it exerts a strength on charged objects in its vicinity.
The Oil-Driblet Experiment
The Oil-Drop Experiment, otherwise known equally the Millikan Oil-Drib Experiment, is i of the most influential studies in the history of physical scientific discipline.
Performed by Robert Millikan and Harvey Fletcher in 1911, the experiment was designed to make up one's mind the charge of a single electron, otherwise known as the elementary electric charge.
Millikan designed his experiment to measure the force on oil aerosol betwixt two electrodes.
He used an atomizer to spray a mist of tiny oil aerosol into a sleeping room, which included a hole. Some droplets would fall through this hole and into a bedchamber, where he measured their terminal velocity and calculated their mass.
Millikan then exposed the aerosol to 10-rays, which ionized molecules in the air and caused electrons to attach to the oil aerosol, thus making them charged. The pinnacle and bottom of the bedroom were attached to a battery, and the potential deviation betwixt the top and bottom produced an electric field that acted on the charged oil drops.
Adjusting the voltage perfectly, Millikan was able to rest the force of gravity (which was exerted downward) with the forcefulness of the electric field on the charged particles (which was exerted upward), causing the oil aerosol to be suspended in mid-air.

Simplified scheme of Millikan's oil-drop experiment: This appliance has a parallel pair of horizontal metallic plates. A uniform electric field is created between them. The ring has three holes for illumination and one for viewing through a microscope. Special oil for vacuum appliance is sprayed into the bedchamber, where drops become electrically charged. The droplets enter the space between the plates and can be controlled by changing the voltage across the plates.
Millikan so calculated the charge on particles suspended in mid-air. His assumptions were that the force of gravity, which is the product of mass (m) and gravitational acceleration (g), was equal to the strength of the electrical field (the product of the accuse (q) and the electric field (E)):
[latex]\text{q}\cdot \text{E}=\text{m}\cdot \text{g}[/latex]
[latex]\text{q}=\frac {\text{m}\cdot \text{g}}{\text{E}}[/latex]
Since he already knew the mass of the oil droplets and the dispatch due to gravity (9.81 m/s^two), too as the free energy of the ten-rays he was using, he was able to calculate the charge.
Although the charge of each droplet was unknown, Millikan adapted the force of the X-rays ionizing the air and measured many values of (q) from many different oil droplets. In each case, the charge measured was a multiple of i.5924(17)×10−19 C. Thus, information technology was ended that the simple electric accuse was 1.5924(17)×10−19 C.
The results were very accurate. The calculated value from the Oil-Drop Experiment differs past less than one percent of the current accepted value of i.602176487(40)×ten−19 C.
The Oil-Drop Experiment was tremendously influential at the time, non simply for determining the charge of an electron, but for helping prove the existence of particles smaller than atoms. At the time, it was non fully accepted that protons, neutrons, and electrons existed.
Source: https://courses.lumenlearning.com/boundless-physics/chapter/overview-2/
0 Response to "How Do You Know Which Way an Electron or Atom Will Point to Electric Fields"
Postar um comentário